The Concept of N-of-1 Trials
Traditionally, in our health system, treatment guidelines and health recommendations have been developed based on results of studies of large groups of individuals. However, the analysis of such large population-based studies only gives insights into factors affecting diseases and treatments on average. Hence, these results do not allow meaningful predictions whether an intervention will help a given single individual. For example, major drugs in therapeutic areas including asthma, depression, diabetes or rheumatoid arthritis have only shown an efficacy in 50%-60% of patients. Similarly, the effect of health interventions and lifestyle changes on chronic disease and health outcomes on an individual level if often unclear.
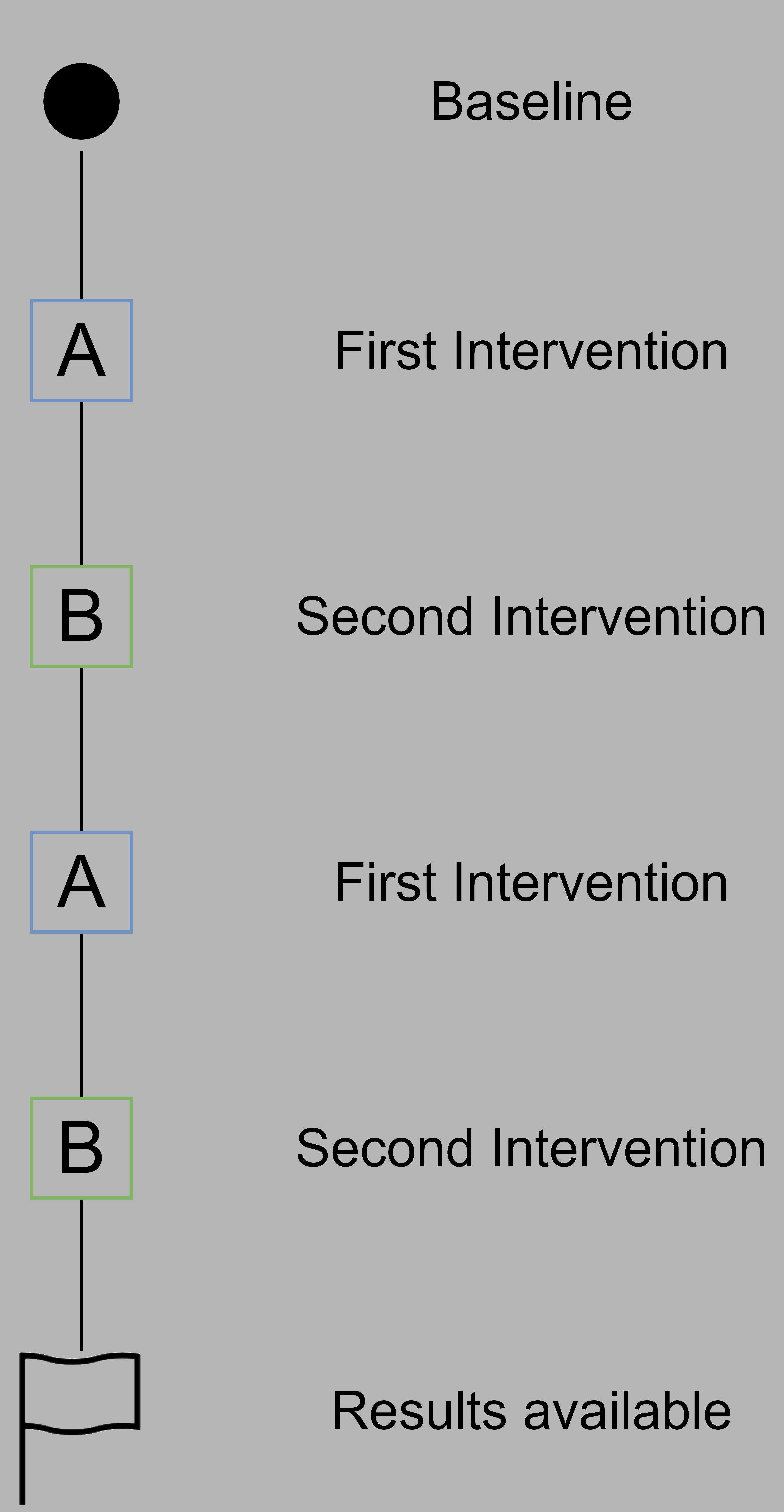
N-of-1 trials provide a framework to evaluate personalized treatments and derive invididual treatment effects. More specifically, N-of-1 trials are multi-crossover randomized controlled trials in a single participant, i.e. where the participant follows the different interventions of the study in a pre-specified or randomized order, and the outcome (e.g. improvement of lower back pain or rheumatoid arthritis pain) is compared between interventions.
If you would like to learn more about N-of-1 trials, we recommend you to watch the short video "What is an N-of-1 trial?" which was created by our collaboration partners at Weill Cornell Medicine.
With StudyU, we are building a platform called StudyU, developing and evaluating necessary tools and methods for the platform, and applying the platform to derive and evaluate individualized treatment effects. Read more about it here.